Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. To facilitate the development of better intervention strategies to counter or prevent tumor development, it is crucial to accelerate the elucidation of the key molecular and cellular mechanisms of oncogenic diseases. As emerging findings highlight the importance of the tumor microenvironment, exploring its contribution to tumor growth and metastasis has become crucial for a better understanding of the molecular and physiological requirements of tumorigenesis.
In recent years, the nematode C. elegans has emerged as a model for systematic dissection of the molecular basis of tumorigenesis. Many cancer genes and pathways are highly conserved and often easier to parse in C. elegans as the gene families involved contain fewer members, reducing genetic redundancy. Several worm models have pioneered the field by helping decipher underlying mechanisms relevant to human tumors such as apoptosis and autophagy. Over the last decade, C. elegans has also been used to advance our understanding of cancer progression, such as deregulation of energy metabolism, stem cell reprogramming, and host-microflora interactions (1).
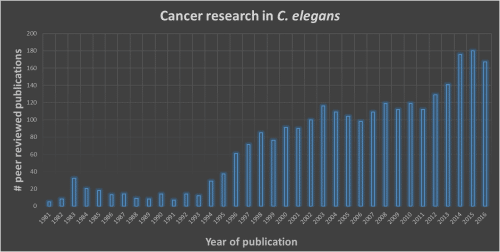 Figure 1: Number of publications using C. elegans a model organism to study cancer mechanisms.
Figure 1: Number of publications using C. elegans a model organism to study cancer mechanisms.
Over the last 2 decades, C. elegans has gained recognition as a suitable model to study the genetic,
molecular and cellular mechanisms of cancer (data from https://www.ncbi.nlm.nih.gov/pubmed)
Several aspects of the C. elegans germ line make it a valuable genetic system for analyzing the cellular and molecular underpinnings of cancer:
- the worm germ line is pluripotent and immortal;
- it is the only C. elegans tissue in which the pattern of apoptosis is not invariant;
- it is the only tissue that undergoes apoptosis in adults.
When the cell cycle and the apoptotic machinery are compromised, the stem cell niche expands, filling the gonad with mitotic nuclei in a way that is similar to tumor development (2, 3). Additionally, many of the genes that modulate tumor growth are orthologs to known human tumor suppressors or oncogenes. Taken together, these traits validate the use of C. elegans as a cancer model.
Researchers have been successfully using C. elegans to:
- Rapidly assess the functional impact of specific gene mutations on tumor development. C. elegans was used to uncover a novel link between hypoxia and apoptosis in tumor progression (4). In addition to apoptosis, C. elegans has contributed significant insights into the role of autophagy in cancer. For example, the pathway in which the tumor suppressor FLCN functions was first delineated in nematodes (5). This study showed that loss of flcn-1, the C. elegans homolog of FLCN, confers resistance to oxidative stress. Autophagy induction, in turn, protects against apoptotic cell death and promotes survival under stress. This pathway was shown to be conserved in mammalian cells, suggesting that FCLN prevents tumor formation by negatively regulating the activity of AMPK (5).
- Assess the outcome of these genes at the organismal level. Pathways that are implicated in mitochondrial repair during mitochondrial dysfunction have been identified in C. elegans. Specifically, a genome-wide RNAi screen revealed 45 genes that are required for mitochondrial repair, detoxification, and pathogen response during compromised mitochondrial function (6). These genes were involved in ceramide and mevalonate metabolism. The mevalonate pathway has been involved in mitochondria-dependent apoptosis in human breast cancer cells (7), and proposed as a target for anticancer therapy (8).
- Screen for new anticancer drugs. The epidermal growth factor receptor (EGFR) is a well-established target for cancer treatment. EGFR is over-expressed or aberrantly activated in various types of human cancer, such as breast, ovarian, and non-small-cell lung carcinoma and is involved in various steps of cancer development. A number of chemical compounds that target the EGFR pathway have been tested using C. elegans vulval development as a model (9,10,11). Using constructed transgenic C. elegans containing several different EGFR constructs, Bae et al. conducted a pilot screen of 8,960 chemicals, isolating an EGFR inhibitor and a MEK inhibitor as suppressors. This study suggest that this C. elegans-based system can be used efficiently to screen for new anti-cancer drugs (12).
| C. elegans Genes | C. elegans Phenotypes | Human Genes | Human Phenotypes | ||
| him gene family(a) | high incidence of males | p53, BRCA1 | cancer (various) | ||
| ksr(b) | aberrant cell specification (multivulva) | RAS family (proto-oncogenes) | improper signal transduction, proliferation and malignant transformation | ||
| Let-60/LIN-45/MEK-, MPK-1(c) | vulva development impairment | RAS/RAF/MEK/ERK | cell proliferation defect during development | ||
| Sep 1 | embryos fail to develop and die at a ertain temperature | ESPL1 | tumorigenesis | ||
| fos-1a | Anchor cell invasion into the vulval epithelium | FOS | metastasis | ||
| mlh-1 | spontaneous mutation rate increased, sterile, embryo lethality | MLH1 | hereditary non-polyposis colon cancer | ||
| mlh-2 | spontaneous mutation rate increased | MSH2 | hereditary non-polyposis colorectal cancer |
- (a) Cancer and the Him phenotype are both the result of chromosome segregation defects.
- (b) Discovery of the ksr gene in D. melanogaster and C. elegans in the mid-1990s has great implications for cancer research.
- (c) The genetic pathways were practically identical: the tyrosine kinase growth factor receptor-Ras-Raf-MEK-MAPK pathway. The same set of orthologs is required for these three biological processes: vulval induction in C. elegans, eye development in D. melanogaster, and cell proliferation in mammalian cell culture.
Conclusion:
C. elegans is widely accepted as a simple model system for elucidating key aspects of stem cell biology (3). The hermaphrodite germ line is the only tissue in C. elegans that can lead to bona fide tumors caused by germline hyper-proliferation and the only tissue capable of undergoing apoptosis throughout adulthood. These characteristics make the worm a straightforward, easily tractable system for studying biological phenomena pertinent to cancer and stem cells.
Unlock the potential of your research with our precise and efficient C. elegans genome-editing services – Contact us now!
See C. elegans genes used as experimental models for investigating other human diseases.
References:
- Kyriakakis, E., Markaki, M. & Tavernarakis, N. Caenorhabditis elegans as a model for cancer research Introducing the Nematode Caenorhabditis elegans: a Compelling Model for the Study of Cancer. doi:10.4161/23723556.2014.975027
- Gartner A, Boag PR,Blackwell TK.Germline survival and apoptosis. WormBook 2008:1-20; PMID:18781708; http://dx.doi.org/10.1895/wormbook.1.145.1
- Joshi PM, Riddle MR, Djabrayan NJ, Rothman JH. Caenorhabditis elegans as amodel for stem cell biology. Dev Dyn 2010; 239:1539-54; PMID:20419785; http://dx.doi.org/10.1002/dvdy.22296
- Sendoel A, Kohler I, Fellmann C, Lowe SW, Hengart- ner MO. HIF-1 antagonizes p53-mediated apoptosis through a secreted neuronal tyrosinase. Nature 2010; 465:577-83; PMID:20520707; http://dx.doi.org/ 10.1038/nature09141
- Possik E, Jalali Z, Nouet Y, Yan M, Gingras MC, Schmeisser K, Panaite L, Dupuy F, Kharitidi D, Cho- tard L, et al. Folliculin regulates ampk-dependent autophagy and metabolic stress survival. PLoS genetics 2014; 10:e1004273; PMID:24763318; http://dx.doi. org/10.1371/journal.pgen.1004273
- Liu Y, Samuel BS, Breen PC, Ruvkun G. Caenorhabditis elegans pathways that surveil and defend mitochondria. Nature 2014; 508:406-10; PMID:24695221; http://dx.doi.org/10.1038/nature13204
- Nakajima, H. et al. Induction of mitochondria-dependent apoptosis through the inhibition of mevalonate pathway in human breast cancer cells by YM529, a new third generation bisphosphonate. Cancer Lett. 253, 89–96 (2007).
- Fritz G. Targeting the mevalonate pathway for improved anticancer therapy. Curr Cancer Drug Targets
- Hara M, Han M (1995) Ras farnesyltransferase inhibitors suppress the phenotype resulting from an activated ras mutation in Caenorhabditis elegans. Proc Natl Acad Sci U S A 92: 3333–3337.
- Gonzalez-Perez V, Reiner DJ, Alan JK, Mitchell C, Edwards LJ, et al. (2010) Genetic and functional characterization of putative Ras/Raf interaction inhibitors in C. elegans and mammalian cells. J Mol Signal 5: 2.
- Lackner MR, Kindt RM, Carroll PM, Brown K, Cancilla MR, et al. (2005) Chemical genetics identifies Rab geranylgeranyl transferase as an apoptotic target of farnesyl transferase inhibitors. Cancer Cell 7: 325–336.13.
- Bae, Y.-K. et al. An In Vivo C. elegans Model System for Screening EGFR-Inhibiting Anti-Cancer Drugs. PLoS One 7, e42441 (2012).